We are surrounded by a number of things. Some of them we can see with our naked eyes and some we cannot see. And each thing is made up of material. So, what is a material? A material is a substance or mixture of substances from which a thing is or can be made. Example: metal, wood, plastics etc. Each material is formed from matter.
Table of Contents
What is the Matter?
The matter is any substance around us that occupies space and has mass. Examples : iron rod, plastic bag, air, water, rocks etc.
What is matter made of?
All matter is made up of very small particles called molecules.
Each molecule of a matter has two or more smaller units called atoms. Atoms are known as building blocks of matter. We cannot see atoms with our naked eyes. When atoms of the same kind come together they form elements. T
To sum up, matter is made up of molecules and molecules consist of atoms.
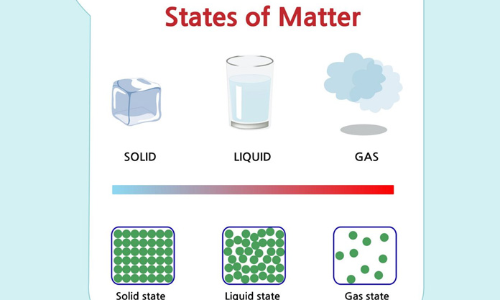
States of Matter
The manner in which the particles of matter are arranged within there are three states of matter known as :
-
Solid
-
Liquid
-
Gas
Molecules are in constant motion and are attracted to each other. Let us see how molecules are arranged in different states of matter.
Molecules in solid :
In solids molecules are very tightly packed together. Thus, the particles cannot flow properly.
Molecules in liquid :
As compared to solids molecules are less closely packed in liquids; so they can move and slide over each other.
Molecules in gases:
In gases, the molecules are at large distance from each other and can move freely.
Solid
Solid is any material that has a fixed shape and volume. The reason for this is because the molecules are tightly packed together which does not allow them to move.
Example:Table, Books, pencils, trees, crystals of sugar etc.
NOTE: we can change the shape of some solids by applying force. For Example: solids like sponge, rubber can change their shapes according to the force applied and removed.
Properties of solid:
- Solids have a definite shape.
- Solids have a definite volume.
- Solids have a definite mass.
- The particles are tightly packed .
- Solids do not flow.
Liquid –
Liquid is any material that does not have a fixed shape. The particles in liquids are loosely packed which allows them to move easily. Thus, they take the shape of the container in which they are poured. But they are packed densely enough that volume is maintained.
Example: Milk, water, oil etc
Properties of liquid :
- Liquids do not have a fixed shape.
- Liquids have a definite volume.
- Liquids have a definite mass.
- The molecules can move freely in liquids. This helps the liquids to flow.
Gas –
Gas is a matter that is composed of very loosely packed articles; therefore they can freely move around. They do not have a fixed shape or volume. It can flow freely and assumes all the space or volume of its container.
Example: air and cooking gas
Properties of gases
- Gases do not have fixed shape and volume.
- A gas does not have a definite mass.
- The molecules in gases are very loosely packed
- Gases can flow easily in all directions.
Dissolving solids, liquids and gases in water
Almost all substances can dissolve in water, making it the universal solvent. Let us see how different states of matter can be combined with water :
1 ) Solids in water :
Some solids get dissolved in water by occupying the space between the molecules of water. For example: sugar and water . However, some substances do not get dissolved in water e.g. chalk powder, sand etc.
2 ) Liquids in water:
Some liquids mix easily when dissolved with water. For example: Alcohol is miscible with water. However, some liquids are insoluble in water e.g. petrol, diesel is immiscible with water.
3) Gases in water:
Some gases get dissolved in water e.g. carbon dioxide, nitrogen, oxygen and ammonia dissolve in water. However, some gases do not get dissolved in water e.g. hydrogen, nitrogen etc.
Difference between the states of matter
| PHYSICAL STATE | SHAPE | VOLUME | FLOW | FORCES OF
ATTRACTION |
SPACE BETWEEN THE PARTICLES |
| Solid | Fixed | Definite | Can not flow | Very strong | Close together |
| Liquid | Not fixed | Definite | Can flow | Intermediate between solids and liquids | Intermediate between solids and liquids |
| Gas | Not fixed | Indefinite | Can flow | Very weak | Intermediate between solids and liquids |
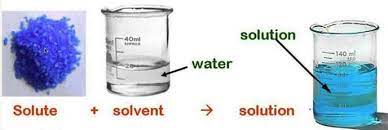
Solution
A solution is a mixture in which substances are completely dissolved. It can be defined as a special type of mixture where two or more substances are combined in such a way that they evenly spread and mix with each other.
Example: sugar mixed with water becomes a solution.
The two substances that make a solution are:
-
Solute
-
Solvent
Solute:
- The substance which is soluble in other substances is called a solute.
- Solute is the component of the solution which is present in small amounts.
Solvent:
- a substance in which the solute is dissolved is called a solvent.
- Solvent is the component of solution which is present in large amounts.
Let us take an example to understand about solute and solvent:
When we mix salt and water it completely dissolves with each other and forms a solution. Here, salt is the solute and water is the solvent.
Types of Solution
There are different types of solutions depending upon the type of solute and solvent present in a solution. They are as follow:
-
Liquid solutions
| SOLUTION | EXAMPLE | SOLUTE | SOLVENT |
| Liquid in gas | Clouds | Liquid(water vapour) | gas(air) |
| Liquid in liquid | Alcohol and water | Liquid(alcohol) | Liquid(water) |
| Liquid in solid | Butter and jam | Liquid | Solid |
-
Solid Solutions
| SOLUTION | EXAMPLE | SOLUTE | SOLVENT |
| Solid in gas | smoke | Solid (carbon particles) | gas(air) |
| Solid in liquid | Sugar water | Solid (sugar) | Liquid(water) |
| Solid in solid | Steel | Solid (carbon) | Solid(iron) |
-
Gas Solutions
| SOLUTION | EXAMPLE | SOLUTE | SOLVENT |
| Gas in gas | Air | Gas(Oxygen) | gas(nitrogen) |
| Gas in liquid | Soft drink | Gas(carbon dioxide) | Liquid(water) |
| Gas in solid | Hydrogen gas on platinum. | Gas(Hydrogen gas ) | Solid(Platinum) |
Soluble and insoluble substances :
Substances that dissolve completely in water to form solutions are termed as soluble substances. For example: Salt, ethanol and sugar are soluble in water.
Substances that do not dissolve in a liquid to form solutions are called insoluble substances. For example: oil,sand, chalk are insoluble in water.
Separation of liquid from Solids
Using different types of methods we can separate soluble and insoluble solutes from solvents. Let us learn more about it .
Separation of soluble substances
We can separate soluble substances from a liquid(e.g; water) by boiling and distillation.
Boiling:
In this process the soluble substances can be separated by heating the water(liquid). When all the water evaporates we will get the substance. Water is lost during the process of boiling.
Example: sugar and water solution is heated till the water evaporates leaving behind sugar particles.
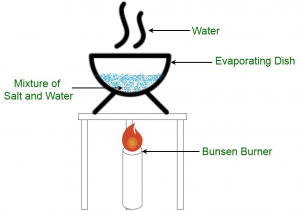
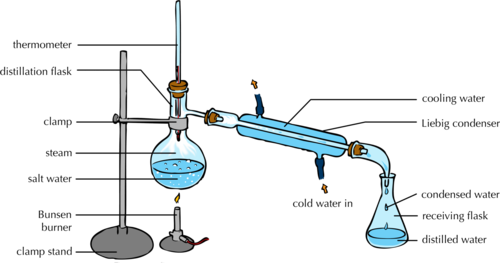
Distillation:
It is a method of purifying water generally performed in laboratories. The water collected after this process is called distilled water. It is free of any impurities. It is used in car batteries and medicines.
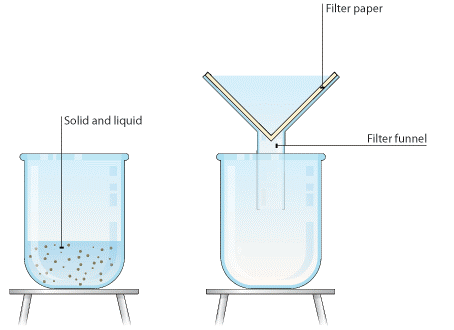
Separation of insoluble substances
Some substances like stones, mud,and sand do not dissolve in liquid(say water) and are called insoluble substances. We can separate the insoluble substances from water by sedimentation followed by decantation and filtration.
Sedimentation and decantation :
When we mix mud in water, the color of the water gets changed. After some time we will see that the insoluble particles of mud settle down at the bottom. These are called sediments. This process of separating insoluble substances is called sedimentation.
Sedimentation steps
- Collect impure water containing mud in a beaker.
- Keep the water undisturbed for some time
- After some time, the mud settles down at the bottom of the beaker and is called sediment.
After the insoluble sediments have settled down, the clear water can be removed into another container. This process is called decantation.
Filtration:
filtration is a process in which water containing insoluble substances are poured into a funnel having a cone of filter paper. Insoluble substances are caught in the filter paper and the clear water passes through it and gets collected in a vessel called a filtrate. This is a better process of separation than sedimentation and decantation.
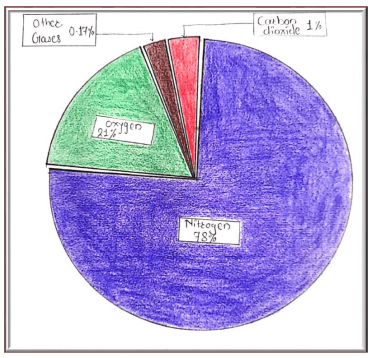
Air
Air is present all around us. Air is an invisible mixture of several gases. It is made up mainly of: 1)nitrogen(78%)
2) Oxygen(21%)
3) Water Vapour
4) 1% of other gases like carbon dioxide, argon, helium, hydrogen etc.
The amount of water vapour present in air determines the humidity of the air.
Formation of wind
Wind can be defined as the movement of air molecules in a large scale. Wind is formed by the uneven heating of the Earth’s surface by the Sun. different surfaces on Earth absorb the Sun’s heat at different rates. As the heat is absorbed by the air, it becomes warm. During this process, warm air rises up as it is lighter than cold air, and cooler air rushes in to replace it. This causes wind.
- Wind: the moving air is called wind.
- Breeze: a gentle wind is called breeze
- Gale: a strong wind is called gale.
- Storm: a very strong and powerful wind with rain is called a storm.
- Thunderstorm: a storm that is accompanied with thunder and lightning is called thunderstorm.
Sea and Land Breezes
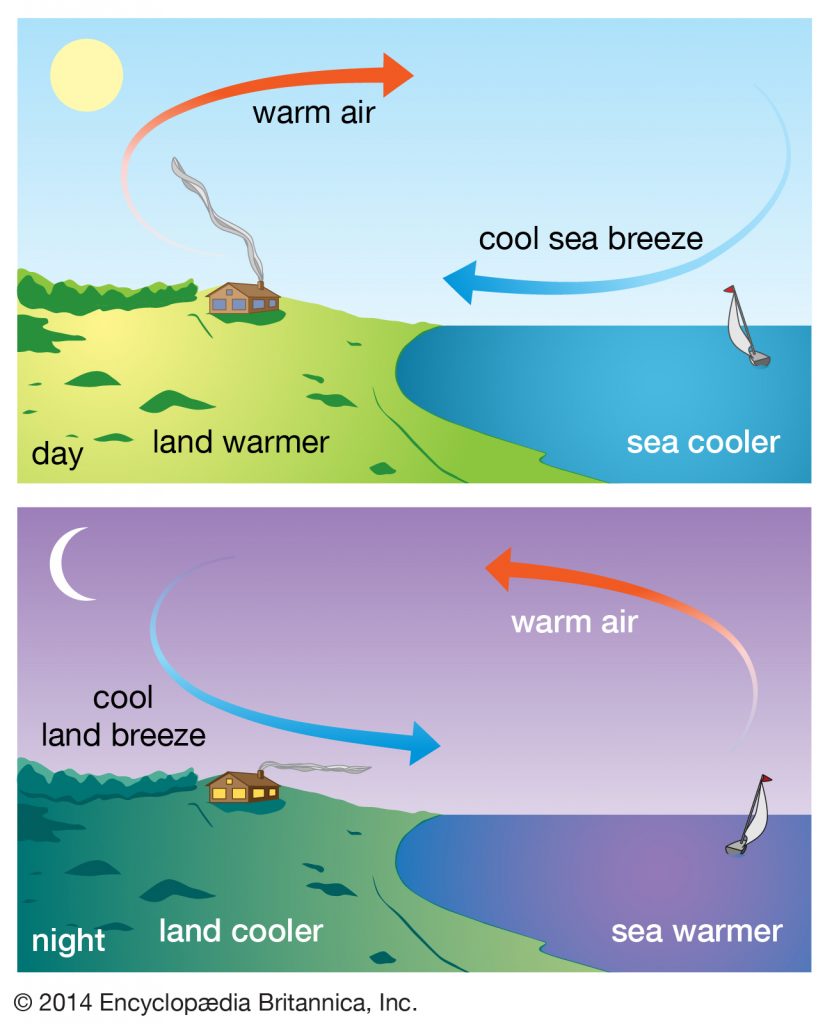
During the day, the land warms up faster than the water. This causes the air above the land to warm up. Warm air being less dense expands and rises leaving an area of low pressure . Meanwhile, the air over the ocean is cooler. The cooler and heavier hair sinks over the water creating an area of high pressure. The difference in pressure and temperature create a convection current. warm air over the land is replaced by the cool ocean air. The cooler wind that blows from the sea to the land is called the sea breeze.
At night, the convection current reverses. After the sun has set the land cools more quickly than water therefore the air above the land also cools down and the air above sea remains warm. The light warm air rises up. The cooler air above the land moves towards the sea to fill up the space. The cooler wind that blows from the land to the sea is called land breeze.
Monsoon Breeze (monsoon wind)
During summers the land near the equator warms up very quickly due to direct sun rays. The temperature of the land is always higher than that of the sea or ocean. The warm land heats air above it as a result of which the warm air being lighter rises up and expands. This causes the moisture -laden air to flow from oceans towards the land . These winds are called the monsoon breeze or wind.
The monsoon wind brings rain in India.
Role of ventilation in closed spaces
We all spend most of our time indoors. The indoor air quality can be many times worse than the air outside. To regulate the flow of fresh air and heat inside the home we use ventilators and exhaust at our homes. Ventilation is simply a process through which we let in the fresh air and take out the bad air. Let us see why ventilation is important:
- It lets fresh air and sunlight come in
- Air pollutants, unpleasant smells also can move out through ventilators.
- As it regulates air flow, it protects our walls and wooden furniture from water damage.
- It keeps our home fresh, dry and airy.
Heating and cooling
Matter can be changed from one state to another with heating or cooling. For example: If we heat ice, it changes into water and its state changes from solid to liquid.
Sometimes heating and cooling bring changes in matter altogether to form a new substance. There are basically two type of changes:
-
Physical change in matter
-
Chemical change in matter

Physical change in matter :
- A physical change is a temporary change which can be reversed.
- No new substance is formed
- Only physical properties of the matter get changed like shape,size, colour etc.
- Chemical properties of the matter remain unchanged.
- For example: Water on cooling becomes ice . Ice on heating becomes water.
Here, only the state of matter is changed which can be easily reversible.
Chemical change in matter:
In chemical change, heating or cooling of matter brings permanent changes in state of the matter.
new substance is formed
This change is irreversible
For example: after we burn a piece of paper it changes into ash. The molecules of paper are different from the molecules of ash.