Table of Contents
What is Matter?
- Matter is anything around us that has the following properties:
- It occupies space.
- It has mass (can be weighed).
- Matter can be found in different forms like solids, liquids, and gases.
Also Check – Rapid Revision –Chapter 1- Matter In Our Surroundings – Class 9 Science
Examples of Matter
- Matter includes:
- Air we breathe.
- Water we drink.
- Food we eat.
- Objects like stones, plants, animals, clouds, and stars.
- Even very small things like a drop of water or a grain of sand are also matter.
What is NOT Matter?
- Feelings like love, anger, thoughts, and smell are not matter because they do not have mass or occupy space.
Characteristics of Matter
- Mass
- Matter has weight, no matter how big or small the object is.
- Example:
- A stone is heavier than a piece of paper because it has more mass.
- Volume
- Matter takes up space.
- Example:
- A glass of water fills the glass completely, showing it occupies space.
Historical Perspective: Panch Tatva and Ancient Classifications
Panch Tatva (Five Elements)
- Early Indian philosophers believed that everything in the world, living or non-living, is made up of five basic elements, called Panch Tatva:
- Air (Vayu)
- Earth (Prithvi)
- Fire (Agni)
- Sky (Aakash)
- Water (Jal)
Ancient Greek Philosophy
- Greek philosophers had a similar idea about matter being made of basic elements.
- Both ideas aimed to explain the diversity of materials in the world.
Also Check – 82 Reasoning-Based Questions & Answers-Matter In Our Surroundings -Class 9 Science
Physical Nature of Matter
Matter is Made Up of Particles
- Matter is not continuous like a block of wood. It is made of tiny particles that are too small to see with our eyes.
Activity to Prove This: Dissolving Salt or Sugar in Water
- Steps:
- Take a glass of water.
- Add a spoonful of sugar or salt to it.
- Stir the water until the sugar/salt disappears.
- Observation:
- The sugar or salt “disappears,” but the water tastes sweet or salty.
- The water level does not rise even after the sugar is added.
- Conclusion:
- Sugar/salt is made of tiny particles that mix with water particles and spread out in the spaces between them.
Particles of Matter are Very Small
- The particles of matter are so small that they cannot be seen directly.
- Even a tiny crystal contains millions of these particles.
Activity to Show How Small They Are: Dilution of Potassium Permanganate
- Steps:
- Take a glass of water and add a few crystals of potassium permanganate (a purple-colored substance).
- Stir until the water turns purple.
- Take 10 mL of this purple solution and mix it with 90 mL of plain water in another glass.
- Repeat this process 5–6 times, each time taking 10 mL of the solution and adding it to 90 mL of water.
- Observation:
- Even after multiple dilutions, the water still has a light purple color.
- Conclusion:
- Just a few crystals contain millions of tiny particles that spread and color the water, proving that particles of matter are incredibly small.
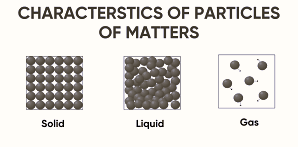
States of Matter
- Matter exists in three main states:
- Solid
- Liquid
- Gas
- The state depends on:
- Arrangement of particles.
- Movement of particles.
- Forces between particles.
The Solid State
Properties of Solids
- Definite Shape and Volume:
- Solids have fixed shapes and occupy a specific volume.
- Example: A book or a pen.
- Rigidity:
- Solids do not flow and maintain their shape.
- Example: A wooden block remains firm.
- Negligible Compressibility:
- Solids cannot be compressed easily because their particles are closely packed.
- Example: A piece of iron does not shrink under pressure.
Special Cases in Solids
- Rubber Bands:
- A rubber band changes shape when stretched but regains its original shape when the force is removed.
- It is considered a solid because it has definite mass and volume.
- Salt and Sugar:
- They take the shape of the container they are in, but their individual particles (crystals) retain their fixed structure.
- Sponge:
- A sponge can be compressed because it has tiny air holes.
- These air pockets get squeezed, but the sponge is still a solid.
Also Check – Chapter 1- Matter In Our Surroundings – Class 9 Science – Long Question and Answers
The Liquid State
Properties of Liquids
- No Fixed Shape:
- Liquids take the shape of the container they are poured into.
- Example: Water in a glass or a bowl.
- Fixed Volume:
- The amount of liquid remains the same, no matter the container.
- Example: 50 mL of milk remains 50 mL whether in a cup or a bottle.
- Fluidity:
- Liquids can flow and be poured.
- Example: Pouring juice from one glass to another.
- Diffusion:
- Liquids can mix with each other or with gases.
- Example: Adding ink to water spreads the color evenly.
Diffusion in Liquids
- Liquids allow gases and other liquids to diffuse into them.
- Example:
- Oxygen dissolves in water, allowing fish to breathe underwater.
- Tea particles mix with water when making tea.
The Gaseous State
Properties of Gases
- High Compressibility:
- Gases can be compressed easily because their particles are far apart.
- Example: Compressed natural gas (CNG) used in vehicles.
- Diffusion:
- Gases mix with other gases quickly.
- Example: The smell of food travels through the air to other rooms.
- Random Movement:
- Gas particles move randomly and spread in all directions.
- Example: Balloons inflate evenly because gas particles spread throughout the balloon.
- Pressure Exertion:
- Gas particles hit the walls of their container, exerting pressure.
- Example: Air pressure in a tire keeps it inflated.
Comparison of Solids, Liquids, and Gases
| Property | Solid | Liquid | Gas |
| Shape | Fixed | Takes shape of container | No fixed shape |
| Volume | Fixed | Fixed | No fixed volume |
| Compressibility | Negligible | Slightly compressible | Highly compressible |
| Flow | Does not flow | Flows easily | Flows and spreads quickly |
| Particle Arrangement | Tightly packed | Loosely packed | Very loosely packed |
Change of State of Matter
- Matter can change from one state to another (solid ↔ liquid ↔ gas).
- The change occurs due to changes in temperature or pressure.
Effect of Temperature
Melting Point
- Melting: When a solid changes into a liquid by heating.
- The melting point is the temperature at which this occurs.
- Example: Ice melts into water at 0°C (273 K).
- What Happens at the Particle Level?
- Heat energy increases the movement of particles, weakening their fixed positions in the solid.
Boiling Point
- Boiling: When a liquid changes into a gas by heating.
- The boiling point is the temperature at which this occurs.
- Example: Water boils into steam at 100°C (373 K).
- What Happens at the Particle Level?
- Heat energy gives particles enough energy to break free from the liquid’s surface into the gaseous state.
Also Check – Chapter 1- Matter In Our Surroundings – Class 9 Science – Solved MCQs
Latent Heat
Latent Heat of Fusion
- The amount of heat energy required to convert 1 kg of a solid into a liquid at its melting point without changing the temperature.
- Example: Ice absorbs heat to melt, but its temperature remains constant at 0°C.
Latent Heat of Vaporization
- The amount of heat energy required to convert 1 kg of a liquid into a gas at its boiling point without changing the temperature.
- Example: Water absorbs heat to become steam, but its temperature remains constant at 100°C.
Also Check – Latent Heat of Fusion Formula- Easy Explanation with Formula & Examples
Also Check – What is Latent Heat? Fusion & Vaporization Explained with Real-Life Applications
Sublimation and Deposition
Sublimation
- A process where a solid changes directly into a gas without becoming a liquid.
- Example: Camphor, naphthalene balls, or dry ice (solid CO₂).
- What Happens at the Particle Level?
- Particles gain enough energy to directly escape into the gaseous state.
Also Check – Sublimation- How Solids Turn Into Gas Without Melting
Deposition
- A process where a gas changes directly into a solid without becoming a liquid.
- Example: Frost formation or solid CO₂ forming under high pressure.
Effect of Pressure
Liquefaction of Gases
- Gases can be converted into liquids by:
- Increasing pressure: Forces particles closer together.
- Decreasing temperature: Reduces particle movement.
- Example: Liquefied Petroleum Gas (LPG) is stored in cylinders by compressing gases under high pressure.
Solid Carbon Dioxide (Dry Ice)
- Solid CO₂ is stored under high pressure.
- On reducing the pressure, it changes directly into gas without becoming liquid (sublimation).
- Used in refrigeration and creating fog effects.
Also Check – NCERT Solutions for Class 9 Science Chapter 1- Matter In Our Surroundings
Key Processes for Change of State
| Process | Change in State | Example |
| Melting | Solid → Liquid | Ice to water |
| Boiling | Liquid → Gas | Water to steam |
| Condensation | Gas → Liquid | Steam to water |
| Freezing | Liquid → Solid | Water to ice |
| Sublimation | Solid → Gas | Camphor, dry ice |
| Deposition | Gas → Solid | Frost forming on surfaces |
Also Chekc – NCERT Exemplar Solutions- Class 9 Science Chapter – 1 – Matter in Our Surroundings
Evaporation
What is Evaporation?
Definition
- Evaporation is the process by which liquid particles at the surface gain enough energy to change into the gaseous state at any temperature below the boiling point.
Also Check – The Science of Evaporation – Process, Factors, and Applications
Phenomenon
- Only Surface Particles Evaporate:
- Particles at the surface of the liquid absorb energy from the surroundings to overcome the forces of attraction and escape into the air as gas.
- Example:
- Water in an open container gradually disappears over time due to evaporation.
Factors Affecting Evaporation
Several factors influence the rate at which evaporation occurs:
1. Surface Area
- Larger surface area increases evaporation.
- More particles are exposed to air, allowing more of them to escape as gas.
- Example:
- Clothes dry faster when spread out compared to being folded.
2. Temperature
- Higher temperature increases evaporation.
- Heat provides more energy for particles to escape into the air.
- Example:
- Water evaporates more quickly on a hot day than on a cold day.
3. Humidity
- Lower humidity increases evaporation.
- Humidity is the amount of water vapor already present in the air.
- If the air is already full of water vapor (high humidity), evaporation slows down.
- Example:
- Clothes take longer to dry on a humid day.
4. Wind Speed
- Higher wind speed increases evaporation.
- Moving air carries away water vapor, leaving space for more liquid particles to evaporate.
- Example:
- Clothes dry faster on a windy day compared to a still day.
Also Check – Matter In Our Surroundings – Worksheet with Answer Key
Evaporation Causes Cooling
How It Happens
- During evaporation, liquid particles absorb energy from their surroundings to overcome the forces of attraction.
- This absorption of energy lowers the temperature of the surroundings, causing a cooling effect.
Examples
- Sweating:
- Sweat evaporates from our skin, taking heat from our body and making us feel cooler.
- Sprinkling Water on the Floor:
- In hot weather, water sprinkled on the floor evaporates, cooling the surface.
- Acetone or Alcohol on the Palm:
- Acetone or alcohol evaporates quickly, absorbing heat from the skin and making the palm feel cold.
Interconversion of States of Matter
- Matter can change from one state to another through specific processes.
- The change depends on conditions like temperature and pressure.
Processes of State Change
1. Melting
- Definition: The process where a solid changes into a liquid on heating.
- Example: Ice melting into water.
- Condition: Occurs at the melting point (e.g., ice melts at 0°C or 273 K).
2. Boiling
- Definition: The process where a liquid changes into gas on heating.
- Example: Water boiling into steam.
- Condition: Occurs at the boiling point (e.g., water boils at 100°C or 373 K).
3. Condensation
- Definition: The process where gas changes into a liquid on cooling.
- Example: Water vapor forming droplets on a cold glass surface.
4. Freezing
- Definition: The process where a liquid changes into a solid on cooling.
- Example: Water freezing into ice.
- Condition: Occurs at the freezing point (e.g., water freezes at 0°C or 273 K).
5. Sublimation
- Definition: The process where a solid directly changes into gas without becoming liquid.
- Example: Camphor, naphthalene balls, and dry ice (solid CO₂) sublimating.
6. Deposition
- Definition: The process where a gas directly changes into a solid without becoming liquid.
- Example: Frost forming on a cold surface.
Conditions Influencing State Changes
1. Temperature
- Effect:
- Increasing temperature provides energy to particles, increasing their movement and causing a change in state (e.g., ice to water).
- Decreasing temperature reduces energy, slowing particle movement and causing condensation or freezing.
- Example:
- Heating water turns it into steam (boiling), while cooling water turns it into ice (freezing).
2. Pressure
- Effect:
- Increasing pressure forces particles closer, changing a gas into a liquid or solid.
- Reducing pressure allows particles to spread, changing a solid into a gas (e.g., sublimation of dry ice).
- Example:
- Liquefied gases like LPG are stored under high pressure to remain liquid.
Key Points to Remember
| Process | Change in State | Example | Conditions |
| Melting | Solid → Liquid | Ice to water | Increase in temperature |
| Boiling | Liquid → Gas | Water to steam | Increase in temperature |
| Condensation | Gas → Liquid | Steam to water | Decrease in temperature |
| Freezing | Liquid → Solid | Water to ice | Decrease in temperature |
| Sublimation | Solid → Gas | Camphor, dry ice sublimating | Increase in temperature or reduce pressure |
| Deposition | Gas → Solid | Frost formation | Decrease in temperature or increase pressure |
Applications of Interconversion
- Refrigeration: Cooling and freezing involve condensation and freezing.
- Cooking: Boiling water and melting ice are examples of temperature-induced changes.
- Industrial Gases: Gases like LPG are liquefied under high pressure for storage and transportation.
Applications and Daily Life Examples
Diffusion in Gases and Liquids
What is Diffusion?
- Diffusion is the process by which particles of matter move from a region of higher concentration to a region of lower concentration.
Examples in Daily Life
- Diffusion in Gases:
- Example: The smell of food or perfume spreads across a room.
- Gases diffuse faster because their particles move freely and are far apart.
- Diffusion in Liquids:
- Example: Adding ink to water spreads its color uniformly.
- Liquids diffuse slower than gases because their particles are closer together.
- Diffusion of Solids in Liquids:
- Example: Sugar dissolving in water is a type of diffusion where particles of sugar mix with water particles.
Importance of Diffusion
- Essential for Life:
- Oxygen diffuses from air into our blood and reaches body cells.
- In aquatic life, oxygen dissolved in water (through diffusion) helps fish and plants survive.
Cooling Effects of Evaporation
How Evaporation Causes Cooling
- During evaporation, particles at the surface of a liquid absorb heat energy from the surroundings to escape into the gaseous state.
- This process removes heat from the surroundings, causing cooling.
Examples in Daily Life
- Acetone (Nail Polish Remover):
- When applied to the skin, acetone evaporates quickly, absorbing heat and making the skin feel cool.
- Desert Coolers:
- Water inside the cooler evaporates, taking heat from the air, and cools the surroundings.
- Sprinkling Water on Hot Surfaces:
- Water evaporates, reducing the heat of the surface.
- Sweating:
- Sweat evaporates from the skin, absorbing body heat and cooling it down.
Role of Diffusion in Survival
Aquatic Life
- Oxygen dissolves in water through diffusion, making it available to fish and aquatic plants for respiration.
Gas Exchange in Plants
- Carbon dioxide diffuses into leaves during photosynthesis.
- Oxygen produced in photosynthesis diffuses out of leaves.
Respiration in Animals
- Oxygen from air diffuses into the bloodstream through the lungs.
- Carbon dioxide diffuses out of the bloodstream into the air for exhalation.
Key Properties and Units
Key Properties and Their SI Units
| Property | Definition | SI Unit | Symbol |
| Mass | The amount of matter in an object. | Kilogram | kg |
| Volume | The space occupied by an object. | Cubic metre | m³ |
| Density | The mass of an object per unit volume. | Kilogram per cubic metre | kg/m³ |
| Temperature | Measure of the average kinetic energy of particles in a substance. | Kelvin | K |
| Pressure | Force exerted per unit area. | Pascal | Pa |
| Length | The distance between two points. | Metre | m |
Comparison of Properties Across States of Matter
| Property | Solid | Liquid | Gas |
| Shape | Fixed | Takes shape of the container | No fixed shape |
| Volume | Fixed | Fixed | No fixed volume |
| Compressibility | Negligible | Very slight | Highly compressible |
| Particle Arrangement | Tightly packed | Loosely packed | Very loosely packed |
| Particle Movement | Vibrates in fixed positions | Moves more freely than solids | Moves randomly at high speeds |
| Interparticle Forces | Strong | Moderate | Weak |
| Diffusion | Very slow | Faster than solids | Very fast |
Examples for Better Understanding
- Solids:
- Example: Iron, wood, ice.
- Particles are tightly packed with strong forces of attraction.
- Liquids:
- Example: Water, oil, milk.
- Particles are less tightly packed and can move past one another.
- Gases:
- Example: Air, carbon dioxide, steam.
- Particles move randomly and have large spaces between them.